In-silico Clinical Trials
In-silico clinical trials are transforming medical device development. Using advanced simulations, devices can be tested in realistic, patient-specific environments before clinical use, accelerating innovation and enabling safer, more efficient, data-driven healthcare solutions.
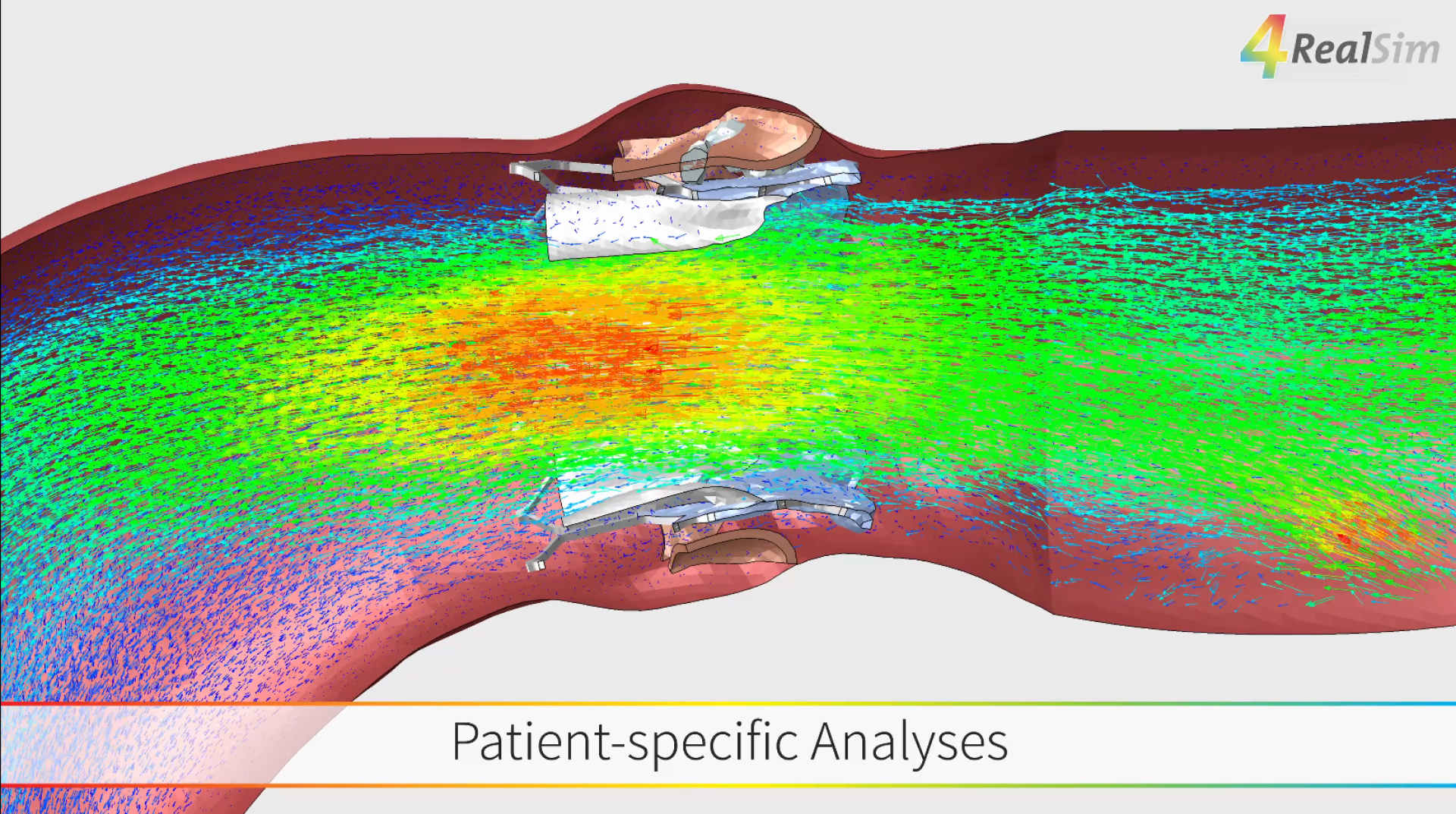
In-silico clinical trials represent an innovative and increasingly important step in the development of medical devices. By leveraging advanced Computer Modelling and Simulation (CM&S), medical devices can be virtually tested within realistic, patient-specific environments. This enables early evaluation of performance, safety, and efficacy—well before traditional clinical studies are conducted.
With growing recognition from regulatory bodies such as the EMA and FDA, in-silico methods are becoming an integral part of the development and validation process, complementing conventional bench testing, in-vitro, and in-vivo studies. Companies like 4RealSim apply high-fidelity multiphysics simulations to support the entire product lifecycle—from early design and feasibility to verification, validation, and ultimately virtual clinical studies.
By combining validated computational models, patient-specific data, and rigorous verification and validation frameworks, in-silico clinical trials offer a powerful alternative or complement to traditional clinical approaches. They enable faster innovation, reduced development costs, and stronger regulatory evidence, ultimately contributing to safer and more effective medical solutions for patients.
Interested in how in-silico clinical trials can accelerate your medical device development? Visit us at our booth to meet our experts and discover how 4RealSim can support your projects. Hall STGT3 Stall 3-360