All-in-one Software for Success in Quality Management
The eQMS suite from Digital Life Sciences GmbH is based on an integrated ECM and DMS system that meets the highest standards of quality and compliance. All software solutions can be expanded in a modular fashion to form a complete GxP-compliant eQMS suite.
What is the eQMS Suite?
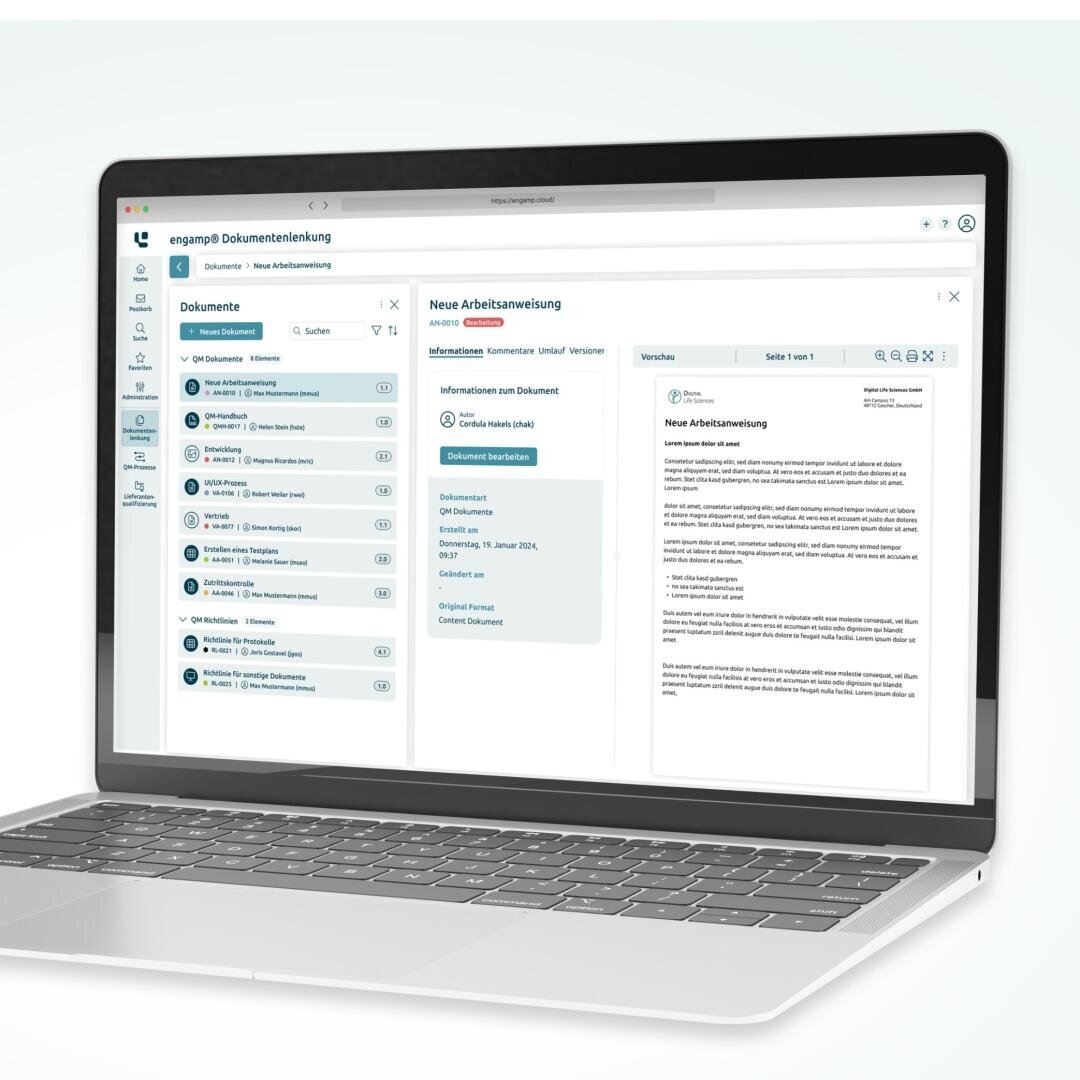
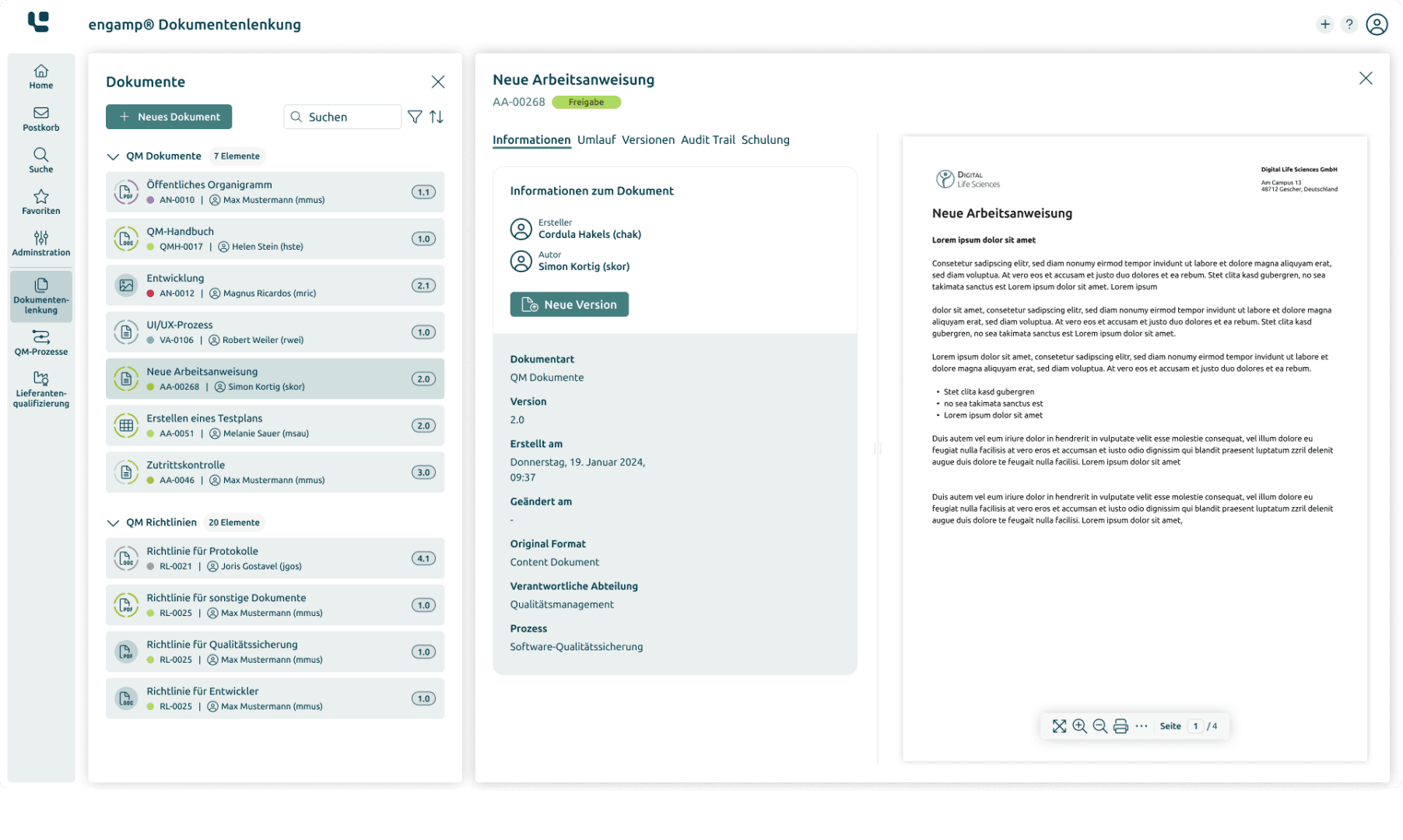
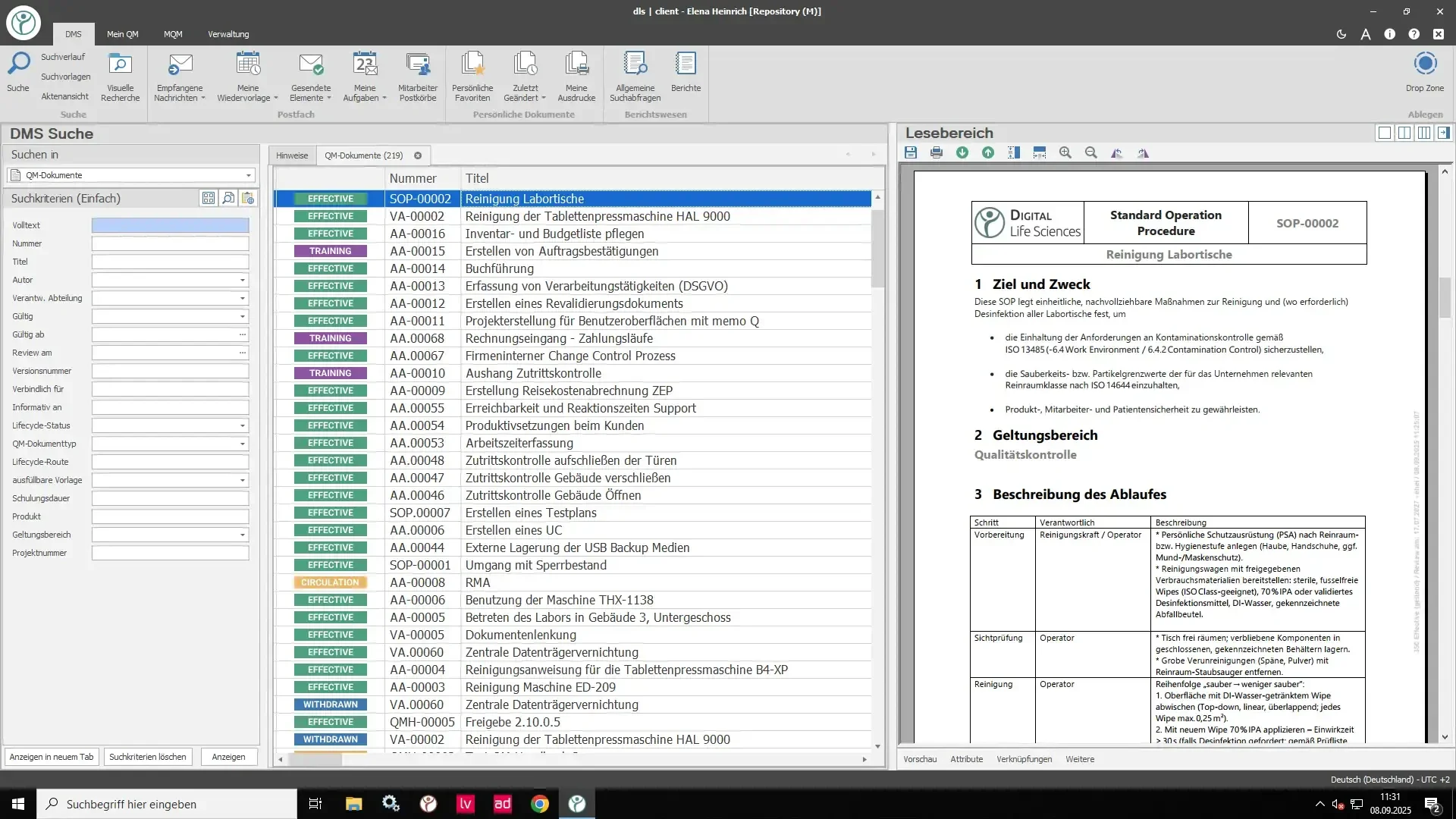
The eQMS Suite was developed specifically for the requirements of the life sciences industry and is based on an integrated document management system (DMS). Our modular software solution allows all modules to be flexibly combined to create a comprehensive, GxP-compliant electronic quality management system (eQMS).
The central modules of the eQMS Suite include:
Document Control: Efficient management and control of documents in accordance with regulatory requirements
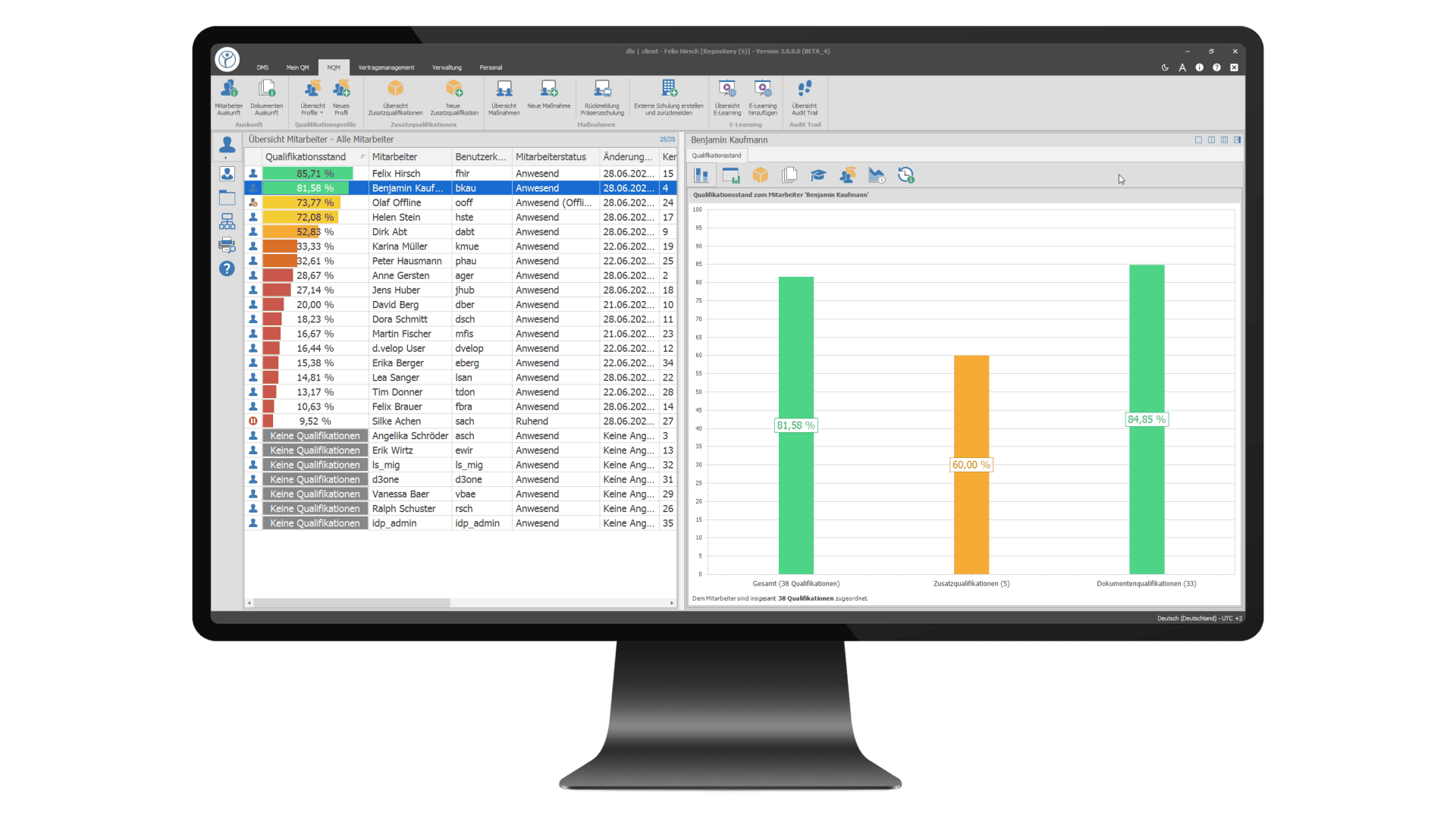
Training Management: Organisation, planning and tracking of training measures
E-learning: Interactive online training courses for employee qualification
Complaint: Systematic recording and processing of complaints
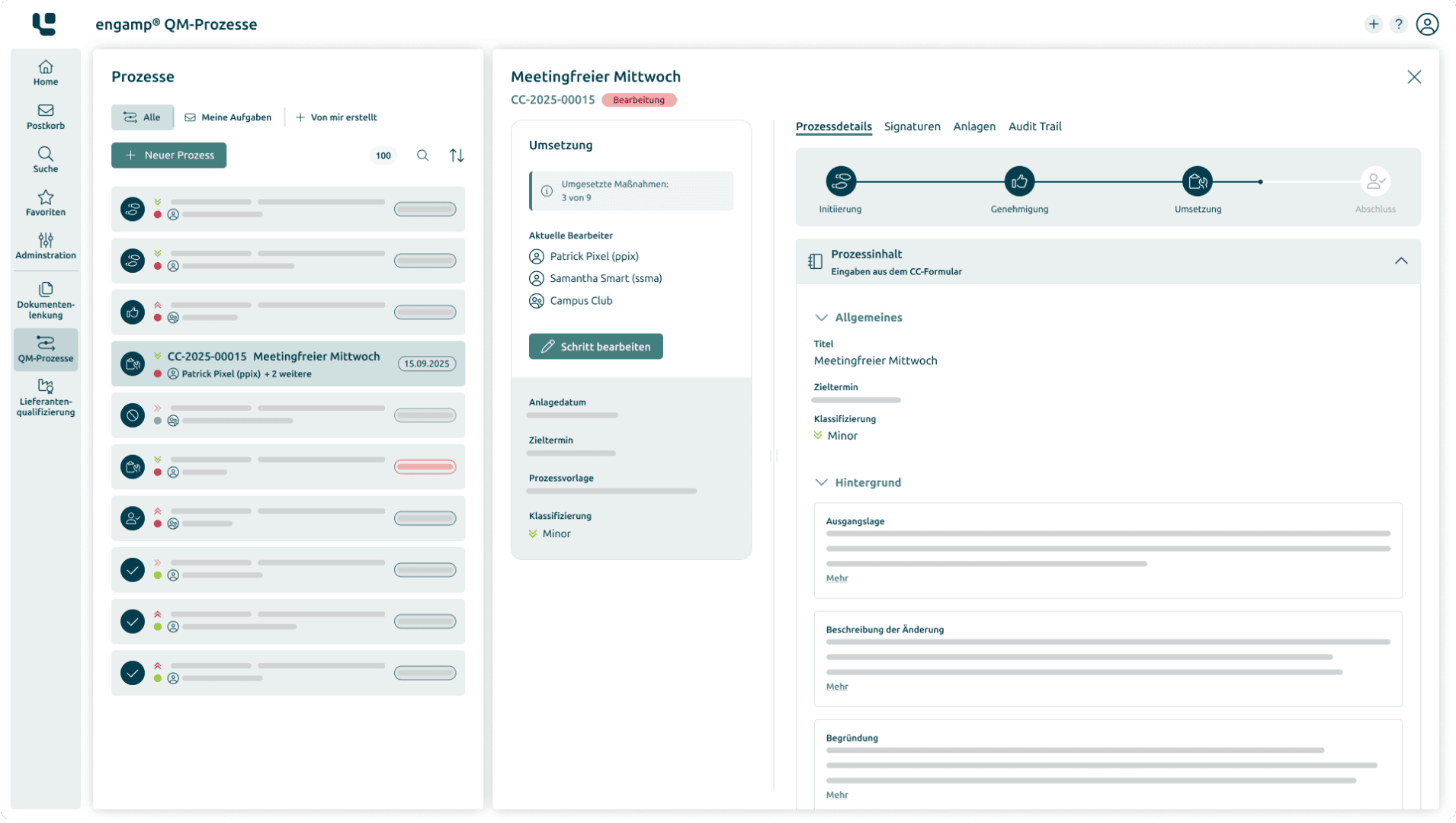
Deviation Control: Systematic recording and processing of deviations
CAPA: Structured implementation of corrective and preventive measures
Change Control: Controlled management and documentation of changes in the system
Technical documentation: Creation and management of technical documents
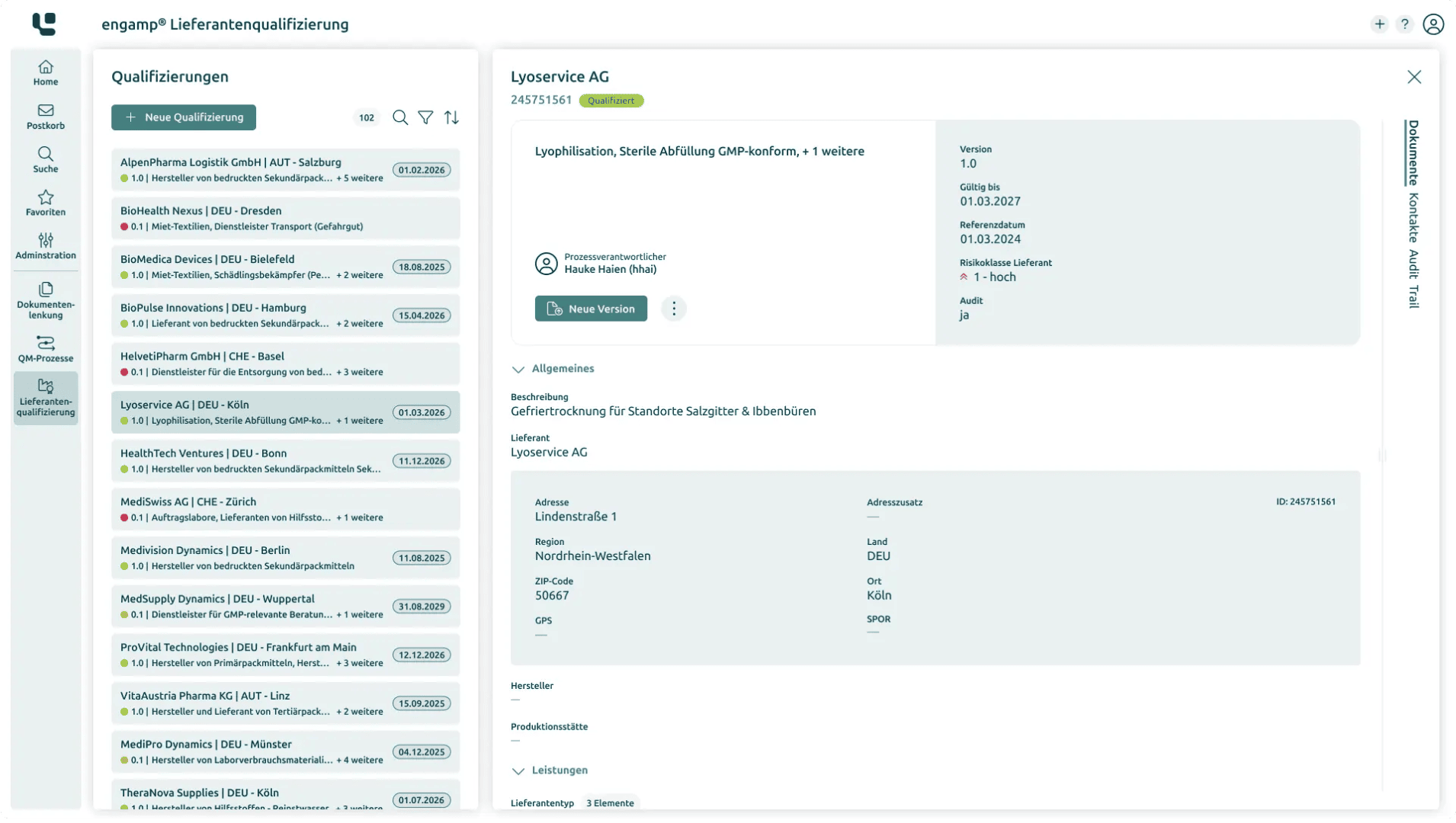
Supplier qualification: Efficient structuring and evaluation of suppliers
Who is the eQMS Suite suitable for?
The eQMS Suite is aimed at companies in the life sciences industry, including pharmaceutical manufacturers, medical technology companies and manufacturers of medical devices, food and cosmetic products. It supports you in complying with GxP guidelines through an integrated solution for managing documents, training, complaints, CAPA measures, change processes and much more. Thanks to its modular architecture, the suite adapts flexibly to your individual requirements and ensures maximum efficiency, transparency and regulatory compliance.