Reduced risk of stroke with the help of 3D printing
Qatna Medical closes the left atrial appendage with an implant to protect against strokes. The operation is successful thanks to the handle made from additively manufactured plastic components from 1zu1. These components mark a milestone for 3D printing and medical technology.
START: From Idea to Medical Concept
Qatna Medical wanted to develop a novel insertion system for a cardiac implant that is minimally invasive, ergonomic, and intuitive to use. The focus was on a sterilizable handle consisting of several components, which requires high precision and must also be suitable for clinical testing and series production.
CHALLENGES: Complexity meets Medical Technology
The project placed the highest demands on design, materials, and manufacturing: undercuts, threads, and narrow channels had to be implemented with precision, while the handle had to remain sterilizable and reproducible. At the same time, close coordination between Qatna Medical and 1zu1scale was necessary to comply with regulatory requirements and ensure ergonomic functionality.
GOAL: Combining Functionality and Feadiness for Series Production
The goal was to develop a functional, regulatory-compliant handle that was ready for series production and met all geometric, ergonomic, and sterilization requirements. The handle was designed to enable safe handling of the cardiac implant during minimally invasive procedures and lay the foundation for future series production.
Innovative Solution for Safe Procedures
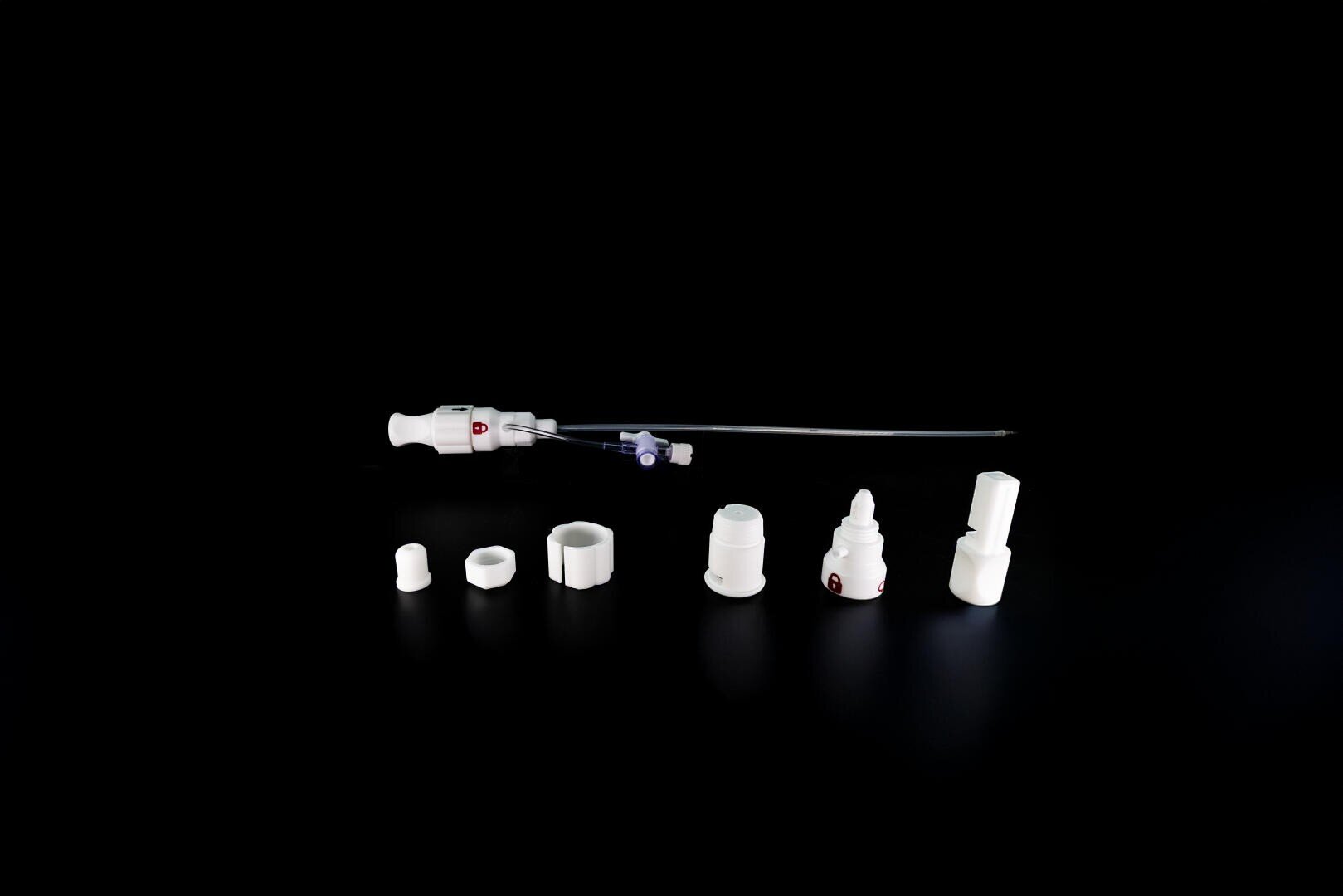
The 3D-printed handle is a key component of the implant delivery system for minimally invasive closure of the left atrial appendage. It enables medical staff to handle the device intuitively and safely during the procedure. The six components are sterile, leak-proof, and easy to clean—crucial factors in a medical environment. Additive manufacturing made it possible to realize complex geometric requirements such as internal threads and underlying channels without expensive tools or complicated assembly. This handle contributes significantly to supporting the vision of improved, low-drug therapy for stroke risk.
SCALE: One Process, many Advantages
S-Sketch: The idea becomes a solution
- Clear definition of requirements right from the start
- Focus on function, biocompatibility, and sterilizability
- Solid foundation for marketable medical technology
C-Check: Trust through validation
- Iterative testing with 3D-printed prototypes
- Realistic test runs and regulatory coordination
- Step-by-step validation until product maturity
A-Approve: Validated. Approved. Finalized.
- Fully validated product design
- Formal approval for the market
- Design freeze as a guarantee for regulatory compliance
L-Launch: Ready for the market
- Structured market launch
- Development of a flexible, additive production strategy
- Ready for series production – scalable, reliable, deliverable
E-Execute: Scaling without compromise
- Series production without product changes
- Flexible market rollout thanks to 3D printing
- Cost-effective manufacturing of complex medical technology – on demand and in series
A Result that is impressive
1zu1scale created a sterilizable handle for Qatna Medical from six 3D-printed components with complex geometries. Additive manufacturing enabled rapid iterations, high geometric freedom, and compliance with medical technology standards. The result: a functional, production-ready component that supports minimally invasive procedures and makes innovative therapy safe for patients.
1zu1scale thinks along with us. Good suggestions, honest, open communication, and face-to-face meetings make it easier for us to implement our ideas and designs. As a developer, this service was a new experience for me. This is exactly how I imagine a solution-oriented manufacturing partner to be.
Felix Schneider, Development Engineer at Qatna Medical GmbH