Correctly labeled for safety: What matters when labeling medical devices
Safety is paramount in medical technology. Laser marking ensures traceability, UDI compliance, and product safety. This article shows how corrosion resistance, material integrity, and validated processes make modern laser technology safe.
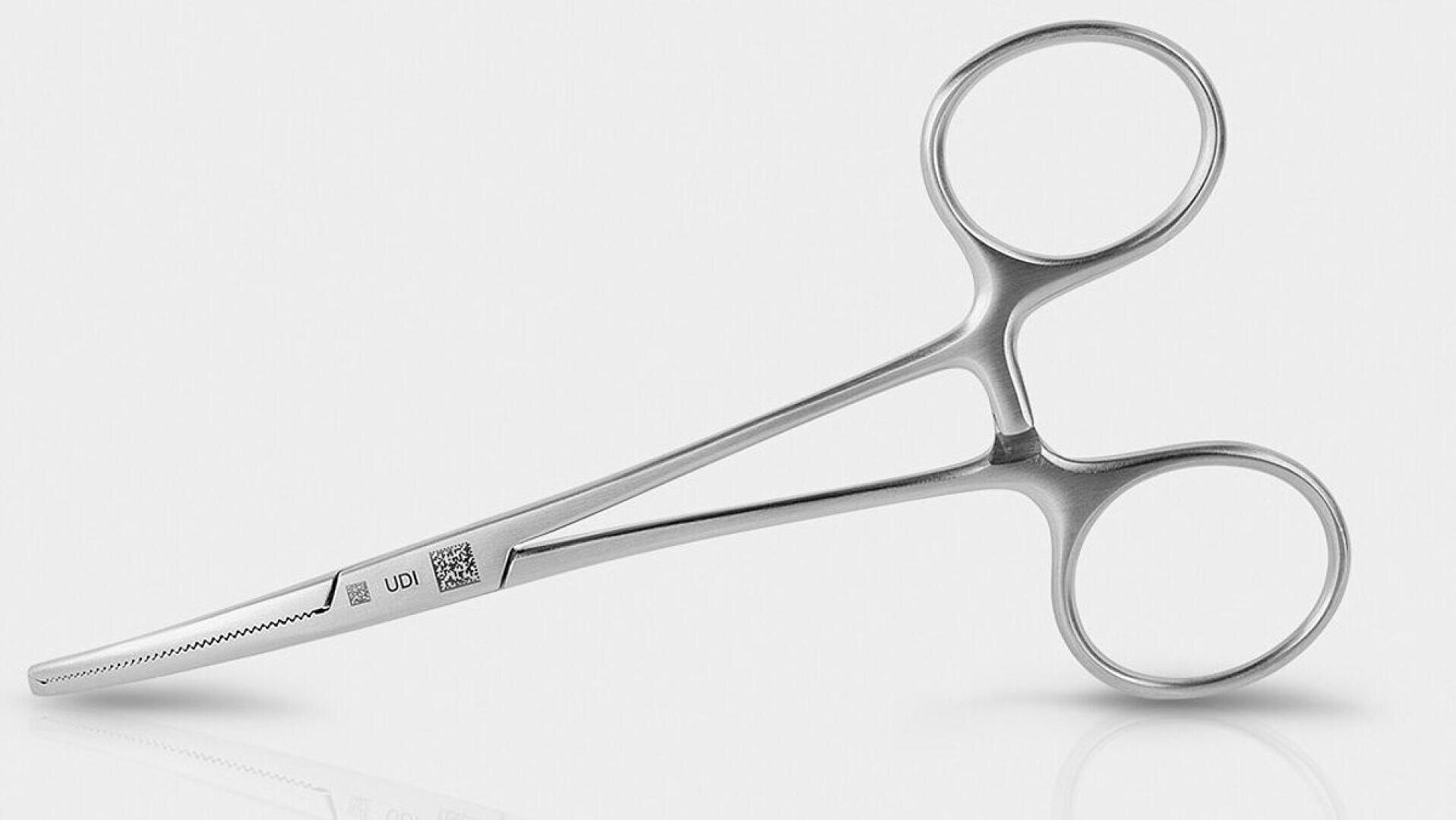
Laser marking is a key element for traceability, UDI compliance, and product safety in medical technology. Decisive factors for the safety of markings include corrosion resistance, material integrity, and the validation of the entire marking process. and the validation of the entire marking process.
Especially with reusable instruments made of stainless steel or titanium, the marking must remain permanently legible and durable even after numerous cleaning and sterilization cycles. For the highest requirements in terms of legibility and surface protection, processes such as black marking with ultra-short pulse lasers or UV laser marking are used. or UV laser marking for sensitive plastics. These technologies enable high-contrast, biocompatible, and abrasion-resistant markings—without thermal stress on the material.
In addition to technology, process reliability is also crucial: camera-based marking processes such as HELP (Holistic Enhanced Laser Process) ensure precise positioning, automatic quality control, complete documentation, and reproducible marking results—decisive criteria for product safety and compliance with regulatory requirements.
Through the combination of expert knowledge, technologically advanced, validated processes, camera-assisted precision, and documented quality, laser technology makes a decisive contribution to product and patient safety.